First steps towards cell therapy for brain disorders
Researchers in Freiburg have found a way to selectively replace defective immune cells in the blood vessels of the brain using an animal model. This is an important first step towards cell therapies for Alzheimer’s disease and other brain disorders. The findings were recently published in the journal Nature Immunology .
The brain and its interfaces are monitored by specialised immune cells. Researchers at the University of Freiburg’s Faculty of Medicine have now investigated macrophages in the meninges and in the brain’s blood vessels, which regulate the removal of waste products from the brain and control inflammatory processes. As scientists from the Institute of neuropathology at the Medical Center – University of Freiburg, in collaboration with an international research team, have demonstrated the brief administration of a drug triggers controlled cell death in these cells. This results in them being specifically replaced by newly arriving cells, thereby influencing their function. This method could form the basis of a new therapy in the future to combat harmful vascular deposits in Alzheimer’s disease. Their research, published in the journal Nature Immunology , was conducted on genetically modified mouse strains. The data obtained are an essential building block for enabling the future use of cell-based therapeutic approaches in brain diseases.
“Using our method, we can selectively replace phagocytes – also known as macrophages – in the blood vessels of the brain. The macrophages within the brain, which are located directly adjacent to the sensitive nerve cells, remain unaffected, however,” says study leader Prof. Dr Marco Prinz , Medical Director of the Institute of Neuropathology at the Medical Center – University of Freiburg and a member of the CIBSS – Centre for Integrative Biological Signalling Studies at the University of Freiburg. “The cells we investigated are all part of the blood-brain barrier, which makes access for therapeutic agents extremely difficult. The fact that we can now, for the first time, specifically replace these macrophages represents a significant step forward,” adds Dr Lukas Amann , group leader at the Institute of Neuropathology at the Medical Center – University of Freiburg and co-last author of the study.
The brain can be divided into two regions: the border zones and the functional tissue. In both, macrophages are the predominant type of immune cell. The macrophages at the brain’s borders are located directly at the blood-brain barrier, which is formed at the brain’s blood vessels. There, they control which substances enter the brain, thereby protecting it from harmful substances, deposits and pathogens. At the same time, waste products are also removed from the brain via the blood vessels, with the macrophages playing an important regulatory role in this process.

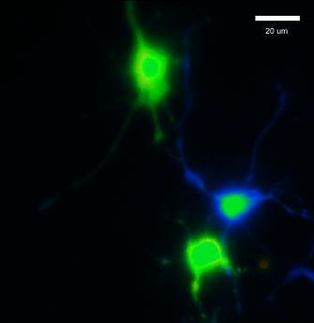
Artistic illustration showing the peripheral regions of the brain, from the dura mater to the parenchyma, with clusters of resident macrophages (blue) and infiltrating macrophages derived from monocytes (yellow).
Universitätsklinikum Freiburg / Maximilian Fliegauf
The implications for Alzheimer’s and other conditions
“Removing waste products from the brain is very important, as these are highly damaging to the brain’s nerve cells. If macrophages are not fully functional, this plays a decisive role in the development of brain diseases. For example, in Alzheimer’s patients, the inadequate removal of harmful proteins from the brain’s blood vessels causes what is known as cerebral amyloid angiopathy. The protein deposits impair the brain’s oxygen supply, which can lead to memory loss and even haemorrhages. We intend to use our findings in future to restore or even improve the function of brain macrophages in such diseases,” Prinz continued.
Glowing immune cells in a stroke model
The research team led by lead author Maximilian Fliegauf , a pharmacist and PhD student at the Institute of Neuropathology at the Medical Center – University of Freiburg, tracked the immune cells in an animal model as they travelled from the bone marrow to the brain and demonstrated that these cells settle long-term specifically in the interfaces of the brain. This was achieved using genetically modified mice in which immune cells were made to glow, allowing them to be observed using high-resolution microscopy. Studies in a stroke model demonstrated that diseases can be influenced by this replacement. “We are demonstrating here for the first time that the immune cells in the brain’s blood vessels can be replaced in a highly targeted manner – and immune cells that can be replaced can also be positively influenced. In the next step, we want to use this knowledge to develop new therapeutic approaches for various vascular diseases of the brain.”
Most read news
Topics
Organizations
Other news from the department science

Get the analytics and lab tech industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.