How oxygen destroys the core of important enzymes
New findings are expected to help protect the hydrogen-producing enzymes from harmful oxygen – which is interesting for biotechnological applications.

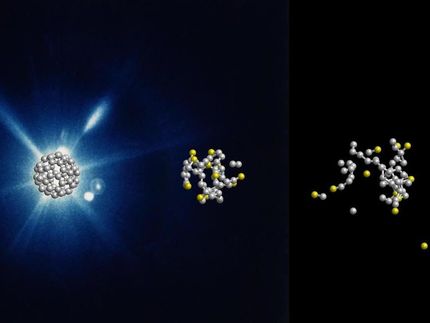
Hydrogenases are very sensitive to oxygen. Thus, experiments with the enzymes have to take place in a sealed environment.
© RUB, Marquard
Certain enzymes, such as hydrogen-producing hydrogenases, are unstable in the presence of oxygen. Researchers at Ruhr-Universität Bochum (RUB) have identified the reasons on the atomic level.
The experiments were carried out jointly by three RUB groups: the photobiotechnology research group was represented by Dr. Julian Esselborn – today at the University of California, San Diego –, Professor Thomas Happe and Dr. Leonie Kertess. The team collaborated with Professor Eckhard Hofmann from the Protein Crystallography Group and Dr. Ulf-Peter Apfel from the Chair of Inorganic Chemistry I.
The interdisciplinary cooperation at the interface between biology, chemistry and physics was embedded in the Ruhr Explores Solvation Cluster of Excellence, Resolv for short, and the Research Training Group Microbial Substrate Conversion, Micon for short.
Structural changes due to oxygen
The researchers analysed a hydrogenase from the bacterium Clostridium pasteurianum. The unique aspect about this class of enzymes is that their structure consists of six iron and six sulphur atoms. The so-called cofactor constitutes the core of the protein, where the actual hydrogen production takes place.
The researchers stored several samples of the enzyme with oxygen for different lengths of time. They then used X-ray structure analysis to study how the three-dimensional structure of the proteins had changed. “This method is very complex and complicated, but it helped us trace the destructive process on an atomic level,” says Julian Esselborn.
Incubation with oxygen only altered individual atoms of the enzyme, namely certain iron atoms of the cofactor. This gradually led to the disintegration of the entire active centre. By understanding which iron atoms are particularly affected, the researchers hope to be able to better protect biotechnologically interesting proteins against oxygen in future.
Original publication
Most read news
Original publication
Julian Esselborn, Leonie Kertess, Ulf-Peter Apfel, Eckhard Hofmann, Thomas Happe; "Loss of specific active-site iron atoms in oxygen-exposed [FeFe]-hydrogenase determined by detailed X‑ray structure analyses"; Journal of the American Chemical Society; 2019
Topics
Organizations
Other news from the department science

Get the analytics and lab tech industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.