Detecting cancer before it spreads
Patrizia Paterlini-Bréchot named European Inventor Award 2019 finalist
The European Patent Office (EPO) announces that cancer researcher Patrizia Paterlini-Bréchot has been nominated as one of three finalists for the European Inventor Award 2019 in the category "Research" for developing a blood filtering technology for the early detection of cancer.
Her invention makes it possible for doctors to detect cancer cells in a blood sample long before a tumour can be found with standard imaging techniques. Importantly, her test can detect these cells before the patient develops metastases - the spread of cancer from the primary tumour to other organs of the body – which is the reason why 90% of patients lose their fight against the disease.
"We know that with cancer treatment effective detection is a key," said EPO President António Campinos, announcing the European Inventor Award 2019 finalists. "Early diagnostic methods such as Patrizia Paterlini-Bréchot's invention are opening up a new front in the battle against this disease by helping doctors to detect the presence of a tumour in its very early stages," he said. "Her story demonstrates how patents can play an important part in driving research that saves and improves lives."
The winners of the 2019 edition of the EPO's annual innovation prize will be announced at a ceremony in Vienna on 20 June.
Inspired by helping patients
There were an estimated 18.1 million new cancer diagnoses worldwide in 2018 and 9.6 million people died of the disease last year. Some cancers are especially difficult to diagnose: lung cancer, for example, is detected at an early stage in only 15% of cases. Finding an effective, non-invasive way to diagnose cancers accurately and early on has the potential to save millions of lives each year. This is also what has motivated the Italian-French researcher throughout her career.
Paterlini-Bréchot studied medicine at the University of Modena, where she specialised in haematology and oncology. After working as a cancer specialist in Bologna, she felt that the most concrete way of helping patients in the long term would be to focus on biomedical research. In 1988 she moved to Paris to train in molecular biology, completing her PhD in 1993 at University of Paris XI on the fundamental bases of oncogenesis. Today Paterlini-Bréchot is Professor of Cellular and Molecular Biology/Oncology at Paris Descartes University, a practitioner in the Paris public hospital system, team research leader at French public research organisation INSERM, and scientific advisor to a company she also founded.
Early on in her career, she lost a cancer patient with an advanced stage of cancer within days of first seeing him. The feeling that she could do nothing to help him shocked her so much that she decided to devote her life to reducing the mortality rate of cancer and to improving patients' lives, something that she explains in her 2017 book Tuer le cancer (Killing Cancer). When asked about this the inventor says: "Often people think that cancer hits a person suddenly and spreads very quickly, but this is not true. What we've learned over the past decades is that the process of forming metastasis is very slow and can go on for years."
Saving lives through early detection
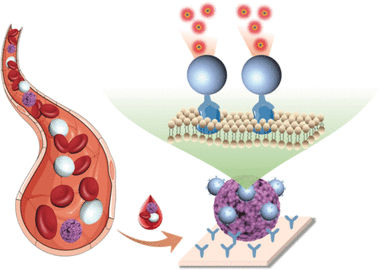
Paterlini-Bréchot decided to take advantage of this "weakness" to fight the disease. She concentrated her research on the possibility of detecting circulating tumour cells (CTCs). These cells, which are shed from the primary tumour and travel through the body in the blood, can be present several years before metastases develop. Since the 1990s they have been a focus of research due to their role in spreading cancer.
Finding CTCs in a patient's blood is a clear sign that tumours are present and that the person is at risk of developing metastasis. The earlier they are detected, the faster and more effectively a patient can be treated. But a number of challenges had to be overcome. For one, CTCs are extremely rare; and they are also incredibly variable. Using her knowledge of oncology and haematology, Paterlini-Bréchot came up with the idea of vertically filtering the blood to isolate CTCs, which is possible due to their larger size compared to other cells. "We phoned all the blood banks at the time to ask if they had a method for vertically filtering blood," she says. "But nobody could help us; they said it was impossible. The problem is that blood clots very quickly and it contains a huge amount of cells. Just 10 milliliters of blood contains on average 100 million white blood cells and 50 billion red ones. The sheer amount clogs every sieve and every filter."
But Paterlini-Bréchot and her team did not give up: they tested more than 700 different ways of filtering blood samples before finally coming up with the right parameters to make it possible to filter and isolate CTCs reliably. She first published work on her new process and device, called ISET® (Isolation by Size of Tumour cells) in 2000, and began filing applications for the first of several European patents.
Filter innovation for better diagnoses
With Paterlini-Bréchot's method blood is first drawn, diluted and then inserted into a small plastic cartridge containing a wafer-thin polycarbonate filter, which works like a coffee filter. The cartridge is inserted into a small table-top machine, which performs the filtration by generating a gentle vacuum that forces the blood sample through the filter.
Smaller blood cells pass through the pores of the filter and, if there are tumour cells in the blood, then these are retained due to their larger size. This process can be completed in about 15 minutes. It is so sensitive that it can detect a single CTC in a 10-millilitre blood sample, or roughly one in about 50 billion blood cells. Cancer specialists can then study cells captured by the filter under a microscope to verify that they are cancer cells, and perform further analysis.
Patients can receive further tests to determine the correct course and timing of therapy, long before metastases occur. In one clinical trial for lung cancer, ISET detected CTCs in patients one to four years before cancerous lung nodules would become visible on CT scans. The test could, therefore, enable doctors to target patients at risk of lung cancer (for example, smokers), in order to make very early interventions if needed.
Furthermore, given that anti-cancer medication targets tumour cells, isolating CTCs in this way can help medical specialists assess whether the treatment is working: anti-cancer drugs that are effective would make CTCs disappear. Another advantage of this method is that the technology can be used to detect virtually all types of solid cancers and even some forms of leukaemia.
Pioneering predictive cancer care
In order to make use of the invention, Paterlini-Bréchot founded Rarecells Diagnostics in 2009. The Paris-based company is a spin-off of Paris Descartes University, INSERM and Assistance Publique - Hôpitaux de Paris, and now holds the exclusive licence to her ISET patents and distributes and develops the tests. The licensing agreement ensures that royalties go back into the public institutions that own the patents. This is an important motivation for Paterlini-Bréchot who believes that research should be for the benefit of the public.
It is also the reason that patent management is such a big priority for her and her team: "It's clear to me that the only way to use research to improve peoples' lives is to bring research products to the market, and patents are essential in this process," the inventor says.
The global CTC diagnostics market was estimated at EUR 8.2 billion in 2017 and is expected to grow to EUR 24.9 billion by 2023. Paterlini-Bréchot is a pioneer in this expanding market. Her ISET test has been available to patients in France since February 2017 – at a cost of just under EUR 500 – as it is currently not covered by health insurance. More clinical trials are needed to establish cancer-specific guidelines for the medical community, a process that requires a great deal of funding. Nonetheless the technology has already been validated by more than 70 independent scientific studies on over 2 000 patients with different types of cancer, and on some 600 individuals without cancer.
Paterlini-Bréchot's next challenge is to identify in which part of the body the cancer originates, as this would facilitate screening, so work continues. Her vision for the future is for ISET to become routine and incorporated into normal check-ups, ensuring that her invention and its benefits are accessible to all.
"Ultimately I think it was good that I went into research," the inventor says. "That way I can help many people at the same time – I can improve and lengthen their lives. If I'd remained a doctor, I'd only have been able to help one patient after the other – rather than all of them at once."