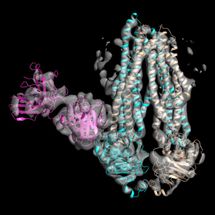
Unlike rubber bands, molecular bonds may not break faster when pulled
From balloons to rubber bands, things always break faster when stretched. Or do they? University of Illinois scientists studying chemical bonds now have shown this isn't always the case, and their results may have profound implications for the stability of proteins to mechanical stress and the design of new high-tech polymers.
"Our findings contradict the intuitive notion that molecules are like rubber bands in that when we pull on a chemical bond, it should always break faster," said chemistry professor Roman Boulatov, who led the study. "When we stretch a sulfur-sulfur bond, for example, how fast it breaks depends on how the nearby atoms move."
The findings also contradict the conventional interpretation of experimental results obtained by other researchers studying the fragmentation rate of certain proteins containing sulfur-sulfur bonds when stretched with a microscopic force probe. In those experiments, as the force increased, the proteins fragmented faster, leading the researchers to conclude that as the sulfur-sulfur bond was stretched, it reacted faster and broke faster.
"Our experiments suggest a different conclusion," Boulatov said. "We believe the acceleration of the fragmentation was caused by a change in the protein's structure as it was stretched, and had little or nothing to do with increased reactivity of a stretched sulfur-sulfur bond."
In their experiments, the researchers use stiff stilbene as a molecular force probe to generate well-defined forces on molecules atom by atom.
The probe allows reaction rates to be measured as a function of the restoring force. Similar to the force that develops when a rubber band is stretched, the molecular restoring force contains information about how much the molecule was distorted, and in what direction.
In previous work, when Boulatov's team pulled on carbon-carbon bonds with the same force they would later apply to sulfur-sulfur bonds, they found the carbon-carbon bonds broke a million times faster than when no force was applied.
"Because the sulfur-sulfur bond is much weaker than the carbon-carbon bond, you might think it would be much more sensitive to being pulled on," Boulatov said. "We found, however, that the sulfur-sulfur bond does not break any faster when pulled."
Boulatov and his team report their findings in a paper accepted for publication in Angewandte Chemie, and posted on the journal's Web site.
"When we pulled on the sulfur-sulfur bond, the nearby methylene groups prevented the rest of the molecule from relaxing," Boulatov said, "thus eliminating the driving force for the sulfur-sulfur bond to break any faster."
Chemists must bear in mind that even in simple chemical reactions, such as a single bond dissociation, "we must take into account other structural changes in the molecule," Boulatov said. "The elongation alone, which occurs when a bond is stretched, does not represent the full picture of what happens when the reaction occurs."
The good news, Boulatov said, is that not every polymer that is stretched will break faster. "We might be able to design polymers, for example, that would resist fragmentation under modest mechanical stresses," he said, "or will not break along the stretched direction, but in some other desired direction."
Most read news
Other news from the department science
These products might interest you

Get the analytics and lab tech industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
Carl Zeiss Ltd. acquires Imaging Associates Ltd.
PerkinElmer and University of Copenhagen Announce Licensing Agreement for New Maternal Health Marker ADAM12
How can we fight blood cancer more effectively? - Several hundred different treatment combinations to be tested simultaneously outside the body
LGC announces plans to expand forensic services with new facilities to be created in Runcorn, Cheshire
Synthetic Production of Potential Pharmaceuticals Dramatically Simplified by Scripps Research Team - Findings Could Expand Interest in Natural Products by Making Production More Cost-Effective
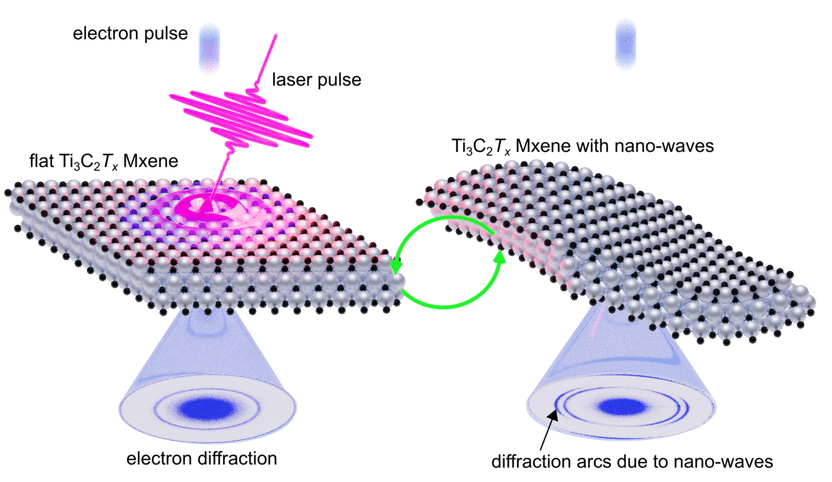
Light-induced shape shifting of MXenes - Femtosecond light creates switchable nano-waves in MXenes and moves the materials’ atoms at a record-breaking speed

Brooks Instrument Joins Major Biotechnology Consortium Focused on Regenerative Drug Manufacturing - Brooks Instrument will contribute insights and precision fluid control technology to the Advanced Regenerative Manufacturing Institute’s BioFabUSA program
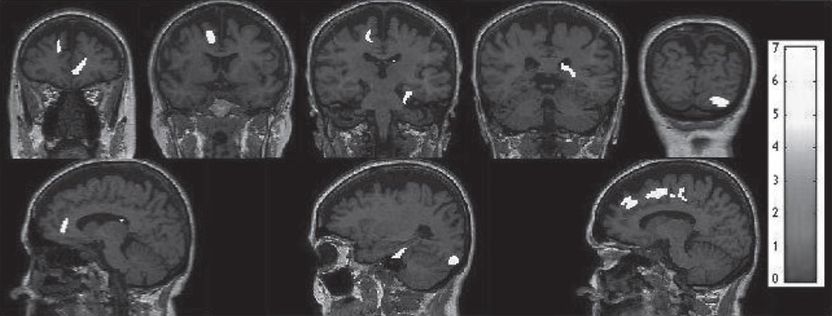
Promising discovery for a non-invasive early detection of Alzheimer's disease

Innovations through hair-thin optical fibres - Study shows what miniaturised optical filters make possible

Wave of the future - Terahertz chips a new way of seeing through matter